Osmosis is the process by which molecules of a solvent pass through a semipermeable membrane from a less concentrated solution into a more concentrated one, equalizing the concentrations on both sides. This movement of molecules is driven by the concentration gradient and does not require energy input. In the context of biology, osmosis is essential for the movement of water and nutrients in and out of cells.
When studying osmosis, it is important to understand key concepts such as hypertonic, hypotonic, and isotonic solutions. A hypertonic solution has a higher solute concentration compared to the cell, causing water to move out of the cell, leading to cell shrinkage. In contrast, a hypotonic solution has a lower solute concentration, causing water to move into the cell, leading to cell swelling. An isotonic solution has an equal solute concentration, resulting in no net movement of water.
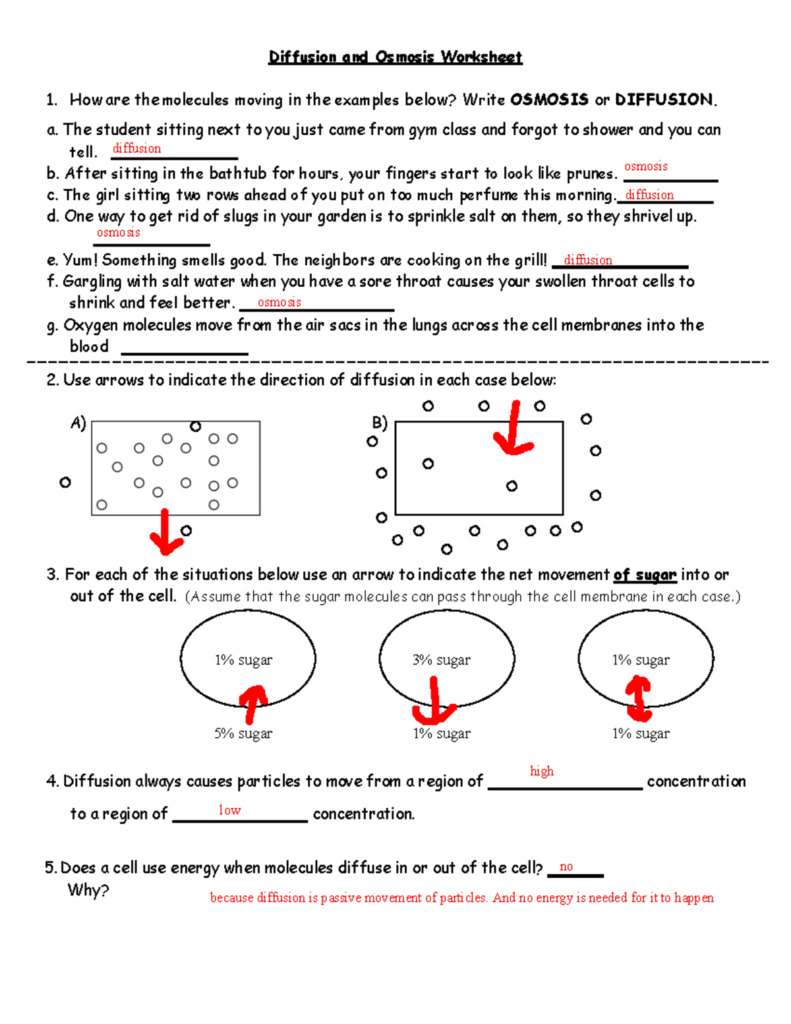
Osmosis Basics Worksheet Answers
Answering Osmosis Basics Worksheet Questions
When faced with osmosis basics worksheet questions, it is crucial to apply the principles of osmosis to solve them accurately. For example, if a question asks about the behavior of a red blood cell in a hypotonic solution, the correct answer would be that the cell would swell due to the influx of water. Similarly, if a question involves calculating the osmotic pressure across a semipermeable membrane, one must consider the concentration gradient and the properties of the membrane.
Worksheet answers for osmosis basics may vary depending on the specific scenario presented in the question. It is essential to review the concepts of osmosis thoroughly and practice applying them to different situations to ensure accurate and confident answers. Additionally, seeking clarification from instructors or using resources such as textbooks and online tutorials can further enhance understanding and mastery of osmosis concepts.