The Plum Pudding Model was proposed by J.J. Thomson in the late 19th century. According to this model, an atom consists of a positively charged sphere with negatively charged electrons embedded in it, much like plums in a pudding. This model helped explain the existence of electrons within an atom and laid the foundation for modern atomic theory.
Thomson’s model was revolutionary at the time but was later replaced by more accurate models such as the Rutherford Model and the Bohr Model. However, understanding the Plum Pudding Model is still essential for students studying atomic structure.
Models Of Atoms Worksheet
The Rutherford Model
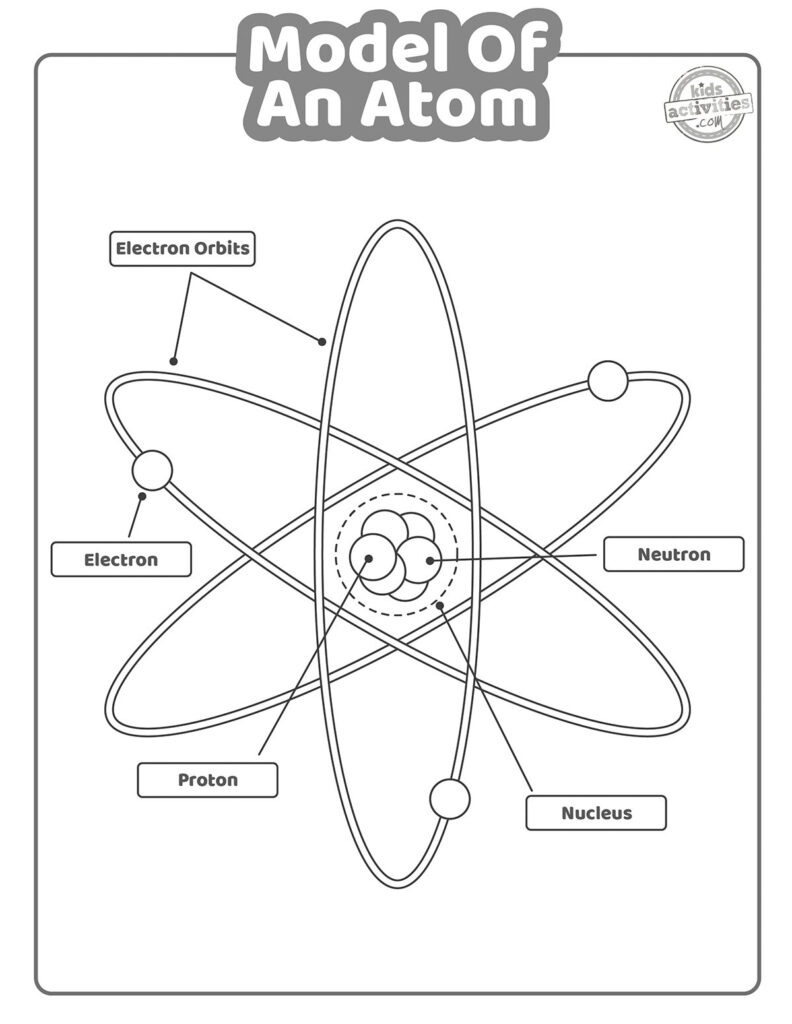
The Rutherford Model, also known as the nuclear model, was proposed by Ernest Rutherford in 1911. This model suggested that atoms have a small, dense, positively charged nucleus at the center, surrounded by orbiting electrons. Rutherford’s famous gold foil experiment provided evidence for this model by showing that most of an atom’s mass is concentrated in the nucleus.
The Rutherford Model marked a significant advancement in our understanding of atomic structure and helped pave the way for further research in quantum mechanics. While it has been refined over the years, the basic concept of a nucleus surrounded by electrons remains a fundamental aspect of modern atomic theory.
The Bohr Model
The Bohr Model, proposed by Niels Bohr in 1913, built upon the ideas of the Rutherford Model and introduced the concept of quantized energy levels for electrons. According to this model, electrons orbit the nucleus in specific, fixed energy levels, or shells, rather than moving in continuous paths.
The Bohr Model successfully explained the spectral lines of hydrogen atoms and provided a more accurate description of atomic structure than previous models. While it has been superseded by more complex quantum mechanical models, the Bohr Model remains a valuable tool for teaching the basics of atomic structure to students.
This article provides an overview of three key models of atoms worksheet: the Plum Pudding Model, the Rutherford Model, and the Bohr Model. Each model offers a unique perspective on atomic structure and has played a crucial role in the development of modern atomic theory. Understanding these models is essential for students studying chemistry and physics, as they provide a foundation for more advanced concepts in the field. By exploring the history and significance of these models, students can gain a deeper understanding of the structure and behavior of atoms at the microscopic level.
Download Models Of Atoms Worksheet
Atomic Models Worksheet Bohr Dalton Rutherford More
Learning The Model Of An Atom Easy Fun Printables Kids Activities Blog
Use This Handwriting Worksheet Generator To Create Models Of Atoms Worksheet